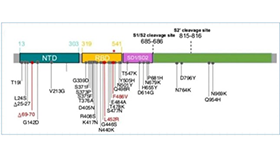
[1]Howard JF Jr, Bril V, Vu T, et al. Safety, efficacy, and tolerability of efgartigimod in patients with generalised myasthenia gravis (ADAPT): a multicentre, randomised, placebo-controlled, phase 3 trial [published correction appears in Lancet Neurol. 2021 Aug;20(8):e5]. Lancet Neurol. 2021;20(7):526-536. doi:10.1016/S1474-4422(21)00159-9.
[2] Chen J, Tian DC, Zhang C, et al. Incidence, mortality, and economic burden of myasthenia gravis in China: A nationwide population-based study. Lancet Reg Health West Pac. 2020;5:100063. Published 2020 Nov 27. doi:10.1016/j.lanwpc.2020.100063.
[3] Gilhus NE, Verschuuren JJ. Myasthenia gravis: subgroup classification and therapeutic strategies. Lancet Neurol. 2015;14(10):1023-1036. doi:10.1016/S1474-4422(15)00145-3.
[4] undefined[M] .Ottawa (ON): Canadian Agency for Drugs and Technologies in Health, 2021.
[5] Howard JF Jr, Utsugisawa K, Benatar M, et al. Safety and efficacy of eculizumab in anti-acetylcholine receptor antibody-positive refractory generalised myasthenia gravis (REGAIN): a phase 3, randomised, double-blind, placebo-controlled, multicentre study [published correction appears in Lancet Neurol. 2017 Dec;16(12 ):954]. Lancet Neurol. 2017;16(12):976-986. doi:10.1016/S1474-4422(17)30369-1.
[6] Muppidi S, Utsugisawa K, Benatar M, et al. Long-term safety and efficacy of eculizumab in generalized myasthenia gravis. Muscle Nerve. 2019;60(1):14-24. doi:10.1002/mus.26447
















.jpg)